73. Nanodiamond Sensing of the Transmetalation Kinetics of Gd-DTPA in Individual Levitated Microdroplets
E. K. Brown, Z. R. Jones, A. Sarkar, B. J. Wallace, A. Ajoy* and K. R. Wilson* [PDF] [SI] ChemRxiv:15001511
Abstract:
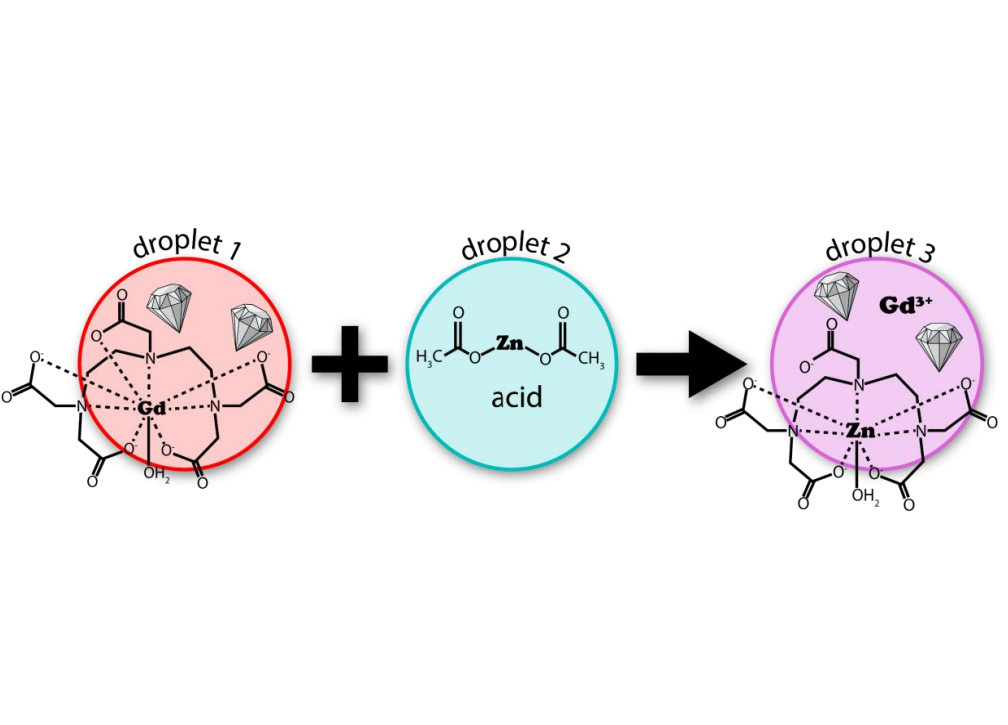
Directly measuring kinetics within microcompartments is critical for determining the distinct mechanisms that govern chemistry in confined environments. Here, we deploy nanodiamond (ND) particles hosting nitrogen-vacancy (NV) centers as quantum sensors to track the transmetalation of gadolinium diethylenetriaminepentaacetic acid (Gd–DTPA) in bulk solution and within single levitated, charged microdroplets by optically detected magnetic resonance (ODMR). The appearance of paramagnetic Gd3+ during reaction produces changes in the continuously acquired ODMR signature, furnishing an in situ optical measurement of kinetics in microscale volumes that are otherwise difficult to interrogate directly. While quantification of [Gd3+] remains challenging due to the high sensitivity of ODMR to pH and ligand concentration, we find that NDs accurately report on observed rate constants for the reaction over a broad pH range by comparison with NMR measurements. A direct comparison between droplet and bulk ND measurements also allows for a reliable kinetic comparison between the two environments. The transmetalation rate in droplets, with an average radius of 18.6 µm, proceeds significantly slower than in a bulk solution, with up to a fourfold reduction in the observed rate constant. Additionally, in the absence of added acid, the transmetalation reaction proceeds efficiently in bulk, yet is completely suppressed in droplets, suggesting that micro-confinement can modulate reactivity. These findings highlight both the strengths and limitations of ND-based ODMR sensing for probing reaction kinetics in microscale environments.